Continuing the Conversation on Lipid Nanoparticle Technologies for Novel Vaccines and Therapies
On April 27th Keystone Symposia hosted a free ePanel event featuring pioneers in lipid nanoparticle design and therapeutic applications, Dr. Philip Felgner, Dr. Terry Allen and Dr. Pieter Cullis. They discussed the evolution of the field from its inception, to the deployment of the first LNP-based immunotherapy, to today, with the advent of mRNA vaccines to against COVID-19.
With 378 registrants from around the world, the event prompted a lively discussion and networking event following. The audience engagement impressive, with an over an overwhelming submission of questions for the panelists. While we were not able to address all of these questions during the live event, our speakers have graciously provided the answers to remaining audience questions below.
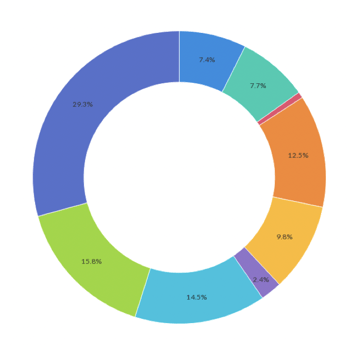
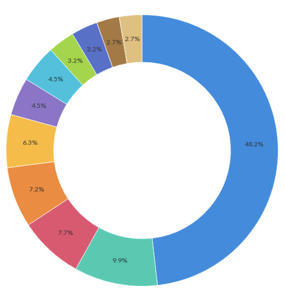
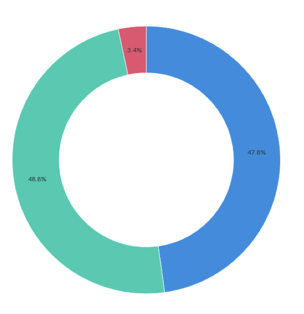
Event Demographics

____________________________________________

____________________________________________

Watch the open access event recording!
Find out more about the panelists on the Keypoint Blog here
The event featured Dr. Cullis as this year's winner of the VinFuture Grand Prize, which honors exceptional minds whose breakthrough scientific research, and technological innovations have enormous potential to create meaningful change at scale.
VinFuture is looking for more high quality nominations for the second season! If you are interested in nominating a potential scientists or a breakthrough scientific and technological invention, submit your nomination below by May 17th, 2022.
Call for VinFuture Prize Nominations
Join us in-person or virtually for the upcoming conference on Vaccines!
Audience Questions
How many RNA molecules can fit in a liposome?
.png?width=192&name=profile-allen-blog%20(1).png)
Dr. Allen: It depends on the diameter of the liposome, and the concentration of the RNA molecules.
.png?width=191&name=profile-Cullis-blog%20(1).png)
Dr. Cullis: Depends on size of the RNA. For LNP siRNA products may have as many as 1,000 siRNAs per LNP. For mRNAs, which are much larger, could be only two or three.
 Dr. Felgner: mRNA LNPs in the Moderna and Pfizer SARS-CoV-2 vaccines have a uniform particle diameter of 70 nm. Each Spike mRNA is about 4000 bases long and is condensed by the positively charged and neutral lipids into a densely packed structure called ‘lipoplex’ and surrounded by a lipid bilayer. There is sufficient volume inside the 70 nm LNP to accommodate ~20,000 RNA nucleotides, 4-6 mRNA Spike molecules. The number of RNA molecules that can be packaged into a 70 nm particle is inversely proportional to RNA length.
Dr. Felgner: mRNA LNPs in the Moderna and Pfizer SARS-CoV-2 vaccines have a uniform particle diameter of 70 nm. Each Spike mRNA is about 4000 bases long and is condensed by the positively charged and neutral lipids into a densely packed structure called ‘lipoplex’ and surrounded by a lipid bilayer. There is sufficient volume inside the 70 nm LNP to accommodate ~20,000 RNA nucleotides, 4-6 mRNA Spike molecules. The number of RNA molecules that can be packaged into a 70 nm particle is inversely proportional to RNA length.
Based on your research, what would be the highest level of tumor accumulation (in terms of ID%) for the PEGylated USVs and the targeted SUVs?
Dr. Allen: Even with PEG and targeting, it would be rare in an in vivo tumor model to get beyond 10% ID, and usually the level of tumor accumulation would be much less.
What are the possible strategies to increase the stability (both in storage and in vivo administration) for liposomes?
Dr. Allen: Various strategies have been used including freeze drying, buffers containing sucrose, low temperature storage, exclusion of oxygen by purging air from vials with Nitrogen, use of Vitamin E (a fat soluble antioxidant), avoiding unsaturated phospholipids to name only a few.
Dr. Cullis: There are a number of possibilities-freeze drying, adding of certain stabilizing excipients, etc. It may be expected that stability of vaccines will improve dramatically for future versions. It may be noted that only very limited stability optimization could be performed for the COVID-19 vaccines due to the urgency of getting these vaccines into the clinic.
Dr. Felgner: In the presence of a simple cryoprotectants like 250 mM sucrose, LNPs can be ‘snap’ frozen at -80 C and thawed numerous times without changing their physical properties or vaccine efficacy, which is convenient for laboratory science. For vaccine applications the LNPs should be recognized by antigen presenting cells at the site of injection or draining lymph nodes within minutes hours after injection.
Vastly HSPC and DSPG are commercially used to make liposomal pharma products. Do we have anything close to these raw materials as alternatives?
Dr. Allen: DSPC is another commonly used phospholipid and also sphingomyelin.
Dr. Cullis: There are many alternatives-fully synthetic lipids such as DSPC as a substitute for HSPC and other versions of PG for DSPG.
Dr. Felgner: At the beginning of the outbreak Avanti Polar Lipids aligned their chemistry and GMP experience with large scale manufacturing from Croda International to produce lipids at the scale necessary to produce billions of vaccine doses and be responsive to this outbreak. There are other lipid manufacturing vendors with similar capability.
Join the panelists at the Liposome Research Days! Jun 12-15, 2022
Jun 12-15, 2022
How to measure PK of liposomes or lipid nanoparticles?
Dr. Allen: These are complex systems with each of the individual components having different pKs.
Dr. Cullis: Measure PK as noted in reference below:
Mui, B. L.; Tam, Y. K.; Jayaraman, M.; Ansell, S. M.; Du, X.; Tam, Y. Y. C.; Lin, P. J.; Chen, S.; Narayanannair, J. K.; Rajeev, K. G. Influence of polyethylene glycol lipid desorption rates on pharmacokinetics and pharmacodynamics of siRNA lipid nanoparticles. Molecular Therapy-Nucleic Acids 2013, 2, e139.
Dr. Felgner: The zetapotential of LNPs can be conveniently measured in the laboratory by dynamic laser light scattering with and instrument called the Zetasizer from Malvern. Depending on the formulation details, LNPs can have a positive, negative or neutral in charge.
Is there any way to distinguish and separate lipoproteins and nanoparticles in the blood stream?
Dr. Allen: Size exclusion chromatograph and sucrose density centrifugation are two methods.
Dr. Cullis: Very difficult to separate lipoproteins and lipid nanoparticles.
Dr. Felgner: In our lab we haven’t yet prioritized separation of LNP from lipoproteins in the blood.
Is there potential to partner nanoparticles with new emerging delivery technologies, such as micro needle array delivery, to increase stability in storage and potentially further increase immunogenicity?
Dr. Allen: A variety of different delivery routes can be used, but the delivery route will affect the rate and location of biodistribution which will, in turn affect the immunogenicity.
Dr. Cullis: Yes, definitely.
Dr. Felgner: Yes. Certainly, the potential of micro needle administration of mRNA vaccines should be investigated. Stability and storage conditions is another area that is under investigation.
What is your vision on the competition between exosomes and liposomes for drug delivery?
Dr. Allen: Liposomes have been in the clinic for decades now for a variety of different applications; exosomes have yet to reach the clinic and more research is need to demonstrate their clinical utility.
Dr. Cullis: Exosomes are too complex and difficult to prepare in a consistent manner.
Dr. Felgner: LNP technology is a leap forward in vaccine manufacturing and regulatory approval. Exosomes can be studied in laboratory and vaccine science to discover vaccine antigens. For pharmaceutical development and regulatory approval, exosomes are heterogenous and challenging to manufacture reproducibly at scale.
Please comment on the pros and cons of using the lipid nanoparticles for neurodegenerative disorders.
Dr. Allen: Lipid nanoparticles are difficult to get across the blood brain barrier and to some other potential sites of biodistribution, so this limits their use, unless their delivery to the sites of action can be improved.
Dr. Cullis: LNP can transfect neurons very well, but it is difficult to get them into the brain
Dr. Felgner: A branch of Alzheimer’s disease science aims to induce an immune response against amyloid plaques. This approach to Alzheimer’s research and development will be another target for mRNA vaccine discovery. See references below:
Transfection agents often release nucleic acids only slowly into the cell once endocytosed - can that delivery be improved?
Dr. Allen: Not sure if there is compelling evidence to suggest that the intracellular release rate is limiting. However these is some evidence to suggest that potency increases when lipids that facilitate the formation of non-bilayer systems, e.g., in endosomes, are used in the nanoparticles.
Dr. Cullis: Yes, that is largely the job of the ionizable cationic lipids. As the pH in the endosome is lowered the ionizable lipid becomes protonated leading to endosome destabilization and release of the nucleic acids into the cytoplasm of the cell. Ionizable cationic lipids are being steadily improved as time goes on.
Dr. Felgner: There is room to improve nucleic acid transfection reagents. Although they can work effectively as a tool for laboratory science, they are not efficient. For example, a typical in vitro DNA transfection exposes each cell to 1 million copies of plasmid, when in theory only 1 plasmid per cell should be necessary. There is a research opportunity to develop LNPs that are more efficient.
Is it possible to create LNPs with microfluidics, has anyone done this?
Dr. Cullis: See reference below.
Belliveau, N. M.; Huft, J.; Lin, P. J.; Chen, S.; Leung, A. K.; Leaver, T. J.; Wild, A. W.; Lee, J. B.; Taylor, R. J.; Tam, Y. K.; et al. Microfluidic Synthesis of Highly Potent Limit-size Lipid Nanoparticles for In Vivo Delivery of siRNA. Mol Ther Nucleic Acids 2012, 1, e37. DOI: 10.1038/mtna.2012.28.
Dr. Felgner: Abe Lee’s lab at UCI has been studying transfection of single cells in a microfluidics platform. See reference below.
What would be the most obstacles for developing and applying nano treatments?
Dr. Allen: One obstacle is finding diseases where a unique mutation can be identified that can cure or controlled the disease when it is amplified or inhibited. Often our common diseases have multiple causes, e.g., heart disease, where a unique cause cannot be identified.
Dr. Cullis: Getting to extrahepatic tissues.
Dr. Felgner: The obstacles for developing and applying LNPs is our lack of basic knowledge. For immunization, we don’t yet understand at a cellular level how antigens encoded on the mRNA are recognized by the immune system. We can make models how we think it is working but lacking data to support the models.
What were the greatest challenges in going from bench to bedside and applying LPNs in the clinic? How were these overcome, and on what timeline? Comments on effective academic-industry collaboration to achieve this?
Dr. Allen: We can, and have, written entire manuscripts and even published books on this, so trying to answer this in a sentence of two is impossible. However, one of the big challenges is finding a clinician champion that can help promote the delivery system so that the large sums of money required for bench to bedside research can be raised. To take the research into the clinic requires funds beyond the resources available to academia and will almost always have to involve industry.
Dr. Cullis: See reference below.
Horejs, C. From lipids to lipid nanoparticles to mRNA vaccines. Nat Rev Mater 2021, 6 (12), 1075-1076. DOI: 10.1038/s41578-021-00379-9 From NLM PubMed-not-MEDLINE
Dr. Felgner: In the ~35 years that it has taken to develop this technology, no one person or one lab could have accomplished what we are experiencing today.
It’s a tribute to the continuity of this scientific enterprise that we are prepared to respond so effectively to this outbreak at this time.
We’re at the dawn of a completely new kind of treatment modality we’ll find integrated into medical practice in the coming years. The worldwide acknowledgment of this triumph provides another opportunity to enjoy the recognition and to encourage the young to help others.
Find out more about Lipid Nanoparticle Vaccines & Therapeutics at These Upcoming Conferences!
____________________________
 Jun 12-15, 2022
Jun 12-15, 2022
Related news
Leveraging Nanoparticle Technologies for Novel Vaccines & Therapies ePanel
Nanoparticles are emerging as a powerful mode of delivery for drugs, vaccines and even gene...
The Evolution of Lipid Nanoparticles: From Biophysics to Gene Therapy, Vaccines and Drug Delivery
On April 27th Keystone Symposia hosted a free ePanel to explore recent breakthroughs in...
KSQA with Dr. Barney Graham on COVID Vaccine Development and Upcoming Keynote Address
The upcoming Keystone Symposia, “Progress in Vaccine Development for Infectious Diseases,” will...